- AdventHealth
On August 3, 2022, AdventHealth Celebration OB/GYN Erica Stockwell, DO, performed the first vaginal-approach hysterectomy in Central Florida using the Anovo™ Surgical System, the first and only FDA-authorized surgical robot designed to enable the minimally invasive transvaginal approach for benign gynecological procedures. Authorized uses include hysterectomy, salpingectomy, oophorectomy, adnexectomy, and ovarian cyst removal.
By incorporating a wrist, elbow and shoulder joint to mimic a surgeon’s movements, Momentis Surgical’s Anovo™ enables a transvaginal approach, providing patients with a quicker return to normal activities compared to abdominal hysterectomy.
“Anovo™ is a hybrid surgical approach that combines the technologic benefits of laparoscopy, like visualization and advanced energy, with the ergonomics of robotics and the optimal patient outcomes of vaginal surgery,” explains Dr. Stockwell.
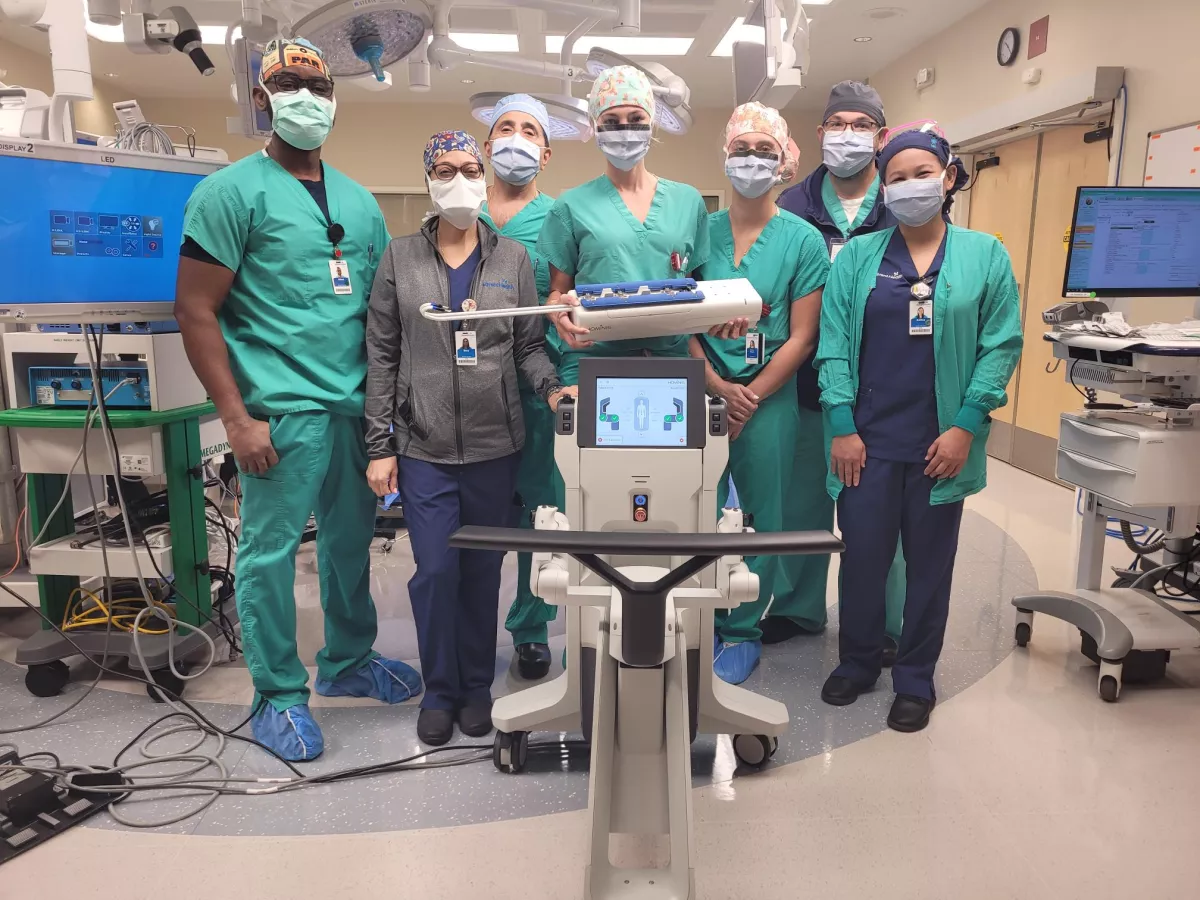
Among U.S. women of reproductive age, hysterectomy is the second most frequently performed surgical procedure, with approximately 600,000 hysterectomies performed each year. In 2017, The American College of Obstetricians and Gynecologists (ACOG) released a statement that vaginal hysterectomy should be the approach of choice whenever feasible because evidence demonstrates that it is associated with better outcomes when compared with other approaches to hysterectomy. However, the less invasive vaginal approach has been used in only 16 percent of hysterectomies due to anatomical and technical limitations. These findings fueled the development of the Anovo™ system.
“We know vaginal hysterectomy provides the best patient outcomes, but poor visualization and access to the pelvis had inhibited its use,” shares Dr. Stockwell. “The Anovo™ system was specifically designed to overcome these barriers and make the vaginal approach more accessible. We believe this new technology will improve care for our patients who require surgery for benign gynecological conditions like abnormal uterine bleeding, fibroids, endometriosis, chronic pelvic pain, prolapse, pre-cancerous changes, and ovarian cysts. It will provide them with less pain and scarring, reduced infection rates, and a quicker recovery and return to their normal daily lives.”
AdventHealth has plans to be to be a primary site for the ongoing registry of clinical outcomes using the Anovo™ system with Dr. Stockwell serving as Primary Investigator.
If you would like to learn more about this procedure, click here.
Clinical Anovo Story
Recent News

AdventHealth Cancer Institute Advances the Use of CAR T-Cell Therapy for Multiple Myeloma
CAR-T therapies have emerged as new treatment options for certain types of cancer through a one-time infusion that can lead to long-lasting remission.

AdventHealth Graduate Medical Education Program Celebrates 50 Years
Growing from a family practice residency to 24 accredited programs, AdventHealth’s Graduate Medical Education (GME) program celebrates 50 years.

New Clinical Trial Explores Digital Intervention for Apathy in Late-life Depression
Afflicting 30-50% of patients with late-life depression, apathy is a challenging psychiatric syndrome in older adulthood that can result in lack of motivation leading to poor self-care, physical...

AdventHealth Performs Central Florida’s First Liver Transplant for Unresectable Colorectal Liver Metastases
In June 2025, AdventHealth Abdominal Transplant Surgeon and Surgical Oncologist Ryan Day, MD, worked with a multidisciplinary team to perform Central Florida’s first liver transplant for unresectable...

AdventHealth Neuroscience Institute First in Florida to be Recognized as a GammaTile Center of Excellence
GammaTile is the first and only tile-based radiation therapy for the treatment of
operable brain tumors. The AdventHealth Neuroscience Institute began performing GammaTile procedures in 2021 and was...

An AdventHealth Collaboration Explores the Impact of Microgravity and Electrical Stimulation on Muscle Cell Health in Space
Using a muscle lab-on-chip model aboard the International Space Station (ISS), AdventHealth Translational Research Institute’s Dr. Paul Coen has been working with a multidisciplinary team from the...

AdventHealth Neuroscience Institute Among First in U.S. to Offer Phase IIa Clinical Trial of Troculeucel for Moderate Alzheimer’s Disease
While most clinical trials for Alzheimer’s disease have focused on patients with early or mild cognitive impairment, roughly 30% of those with Alzheimer’s have moderate stage disease for which there...

AdventHealth Translational Research Institute Selected as Clinical Site for National Study of Muscle, Mobility and Aging (SOMMA)
Under the leadership of Scientific Director and Principal Investigator Bret H. Goodpaster, PhD, the AdventHealth Translational Research Institute (TRI) is now enrolling men and women 70 years of age...

AdventHealth Neuroscience Institute Administers First Dose of Investigative NK Cell Therapy to Person with Alzheimer’s Disease
Under a single compassionate use Investigational New Drug (IND) authorization cleared by the U.S. Food and Drug Administration (FDA), the AdventHealth Neuroscience Institute worked with NKGen Biotech...
New Study Published in the New England Journal of Medicine Shows the Addition of Regional Nodal Irradiation Does Not Decrease Rate of Invasive Breast Cancer Recurrence in Patients with Negative Axillary Nodes Following Neoadjuvant Chemotherapy
Findings of the phase III, multicenter, randomized NSABP B-51-RTOG 1304 clinical trial were recently published in the New England Journal of Medicine with AdventHealth Cancer Institute’s Dr. Mamounas...

AdventHealth Study Exploring the Use of MR-Guided Focused Ultrasound (MRgFUS) to Disrupt the Blood-Brain Barrier for Treatment of Alzheimer’s Disease
Under the leadership of Dr. Valeria Baldivieso and Dr. Chandan Reddy, the AdventHealth Research Institute is the first and only site in Orlando offering the Exablate Blood-Brain Barrier (BBB)...

AdventHealth Research Institute Offers Phase II Clinical Trial of Etanercept for Treatment of Blast-Induced Tinnitus
Approximately 1 billion people throughout the world suffer from tinnitus (ringing in the ears), and it is the number one disability of those who serve in the U.S. military. The condition can disrupt...
